Digoxin, an approved medication for certain heart conditions, promoted the repair of myelin — the protective sheath around nerve fibers that is progressively lost in multiple sclerosis — in mouse models of the disease, a study reports.
Combining digoxin with an experimental immune-modulating therapy was more effective at promoting myelin repair, or remyelination, than either treatment on its own.
“We are planning a phase 1 trial of digoxin in MS patients with currently approved disease-modifying therapies,” the researchers wrote.
The study, “Repurposing the cardiac glycoside digoxin to stimulate myelin regeneration in chemically-induced and immune-mediated mouse models of multiple sclerosis,” was published in Glia.
MS is caused by abnormal immune attacks that damage the myelin sheath, a fatty coating around nerve fibers, or axons, that help speed the transmission of signals between nerve cells.
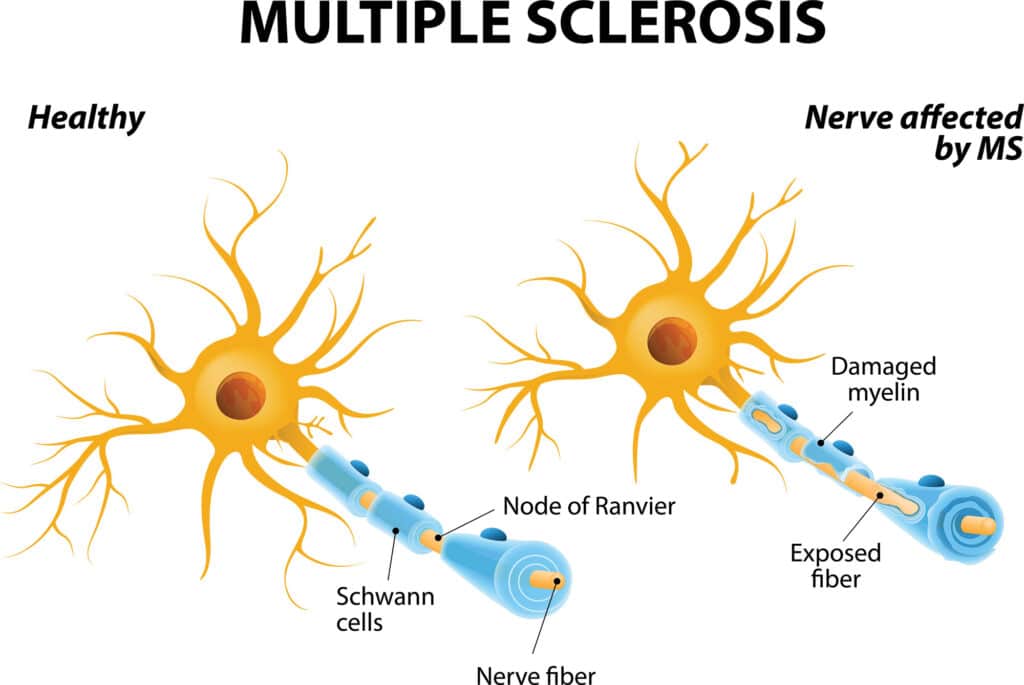 Available MS treatments work broadly by suppressing immune responses and inflammation, but there are no approved therapies that can promote the repair of damaged or lost myelin — a process that is deficient in MS.
Available MS treatments work broadly by suppressing immune responses and inflammation, but there are no approved therapies that can promote the repair of damaged or lost myelin — a process that is deficient in MS.
To accelerate the discovery of molecules with remyelinating potential, researchers have conducted several high throughput screens of treatments already approved for other conditions.
Remyelination potential seen in EAE mouse model
One such screening suggested that digoxin, a medication approved for heart failure and irregular heartbeat, may have remyelination-promoting properties. Sold under the brand name Lanoxin, digoxin also is available in generic forms.
A research team in the U.S. conducted a series of tests to explore digoxin as a potential remyelinating therapy.
The researchers first examined the medication’s effect on oligodendrocytes, which are the cells chiefly responsible for making myelin in the brain and spinal cord. Experiments done in lab dishes indicated that treating immature oligodendrocyte progenitor cells (OPCs) with digoxin prompted these cells to mature into myelin-making oligodendrocytes.
They next tested digoxin, administered via injections into the abdomen, in two mouse models of chemically-induced myelin loss, or demyelination. In both models, treatment with digoxin robustly increased remyelination, and this increase was accompanied by a raise in mature oligodendrocyte counts.
Notably, “the initial changes in myelin regeneration were present prior to a significant change in oligodendrocyte cell number,” the researchers wrote.
This suggests that digoxin promotes myelin repair by “restabilizing [oligodendrocyte] function in existing mature cells and/or promoting the natural turnover of dysfunctional [oligodendrocytes] and maturation of the OPC population,” the team added.
To examine digoxin in a more clinically relevant model, the researchers tested the medication in mice with experimental autoimmune encephalomyelitis, or EAE, which is commonly used as a mouse model of MS.
Results showed that an extended 30-day treatment with digoxin, but not a short-term regimen, significantly increased the number of myelin-producing oligodendrocytes and eased symptom severity in this model.
Researchers also tested digoxin in combination with an experimental therapy designed to promote immune tolerance toward myelin. Called PLG-MOG, the approach uses nanoparticles made of a biodegradable molecule called PLG to deliver a fragment of MOG, a key myelin protein, to immune cells.
“Digoxin and [PLG-MOG] are ideal for combination therapy because of their independent mechanisms of action, ameliorating different steps of the disease process and mutual potentiating effects,” the team wrote.
A single dose of PLG-MOG, alone or in combination with digoxin, was found to substantially reduce symptom severity in mice with EAE, and to a greater extent than digoxin alone.
The combination therapy also significantly increased the numbers of OPCs and myelin-producing oligodendrocytes, “which was not observed with either individual treatment,” the researchers wrote.
These data show “combining targeted myelin tolerance immunotherapy, thus avoiding the serious side effects of immunosuppressive therapies, with myelin repair/regenerative therapy is highly effective in ameliorating [MS-like disease], exceeding the effects of either therapy alone,” they added.
“These findings provide critical pre-clinical evidence supporting future clinical trials of myelin-specific tolerance with myelin repair/regeneration drugs, such as digoxin, in MS patients,” the researchers wrote.
The team is planning a Phase 1 trial of digoxin in MS patients on approved disease-modifying therapies.
One of the study’s authors is a co-founder of Cour Pharmaceuticals, which is developing a PLG-MOG-like approach as a treatment for early relapsing-remitting MS.
What are your thoughts on this study?
Let us know in the comments below!
What topics should we cover next?
Email us your ideas at info@painresource.com!
Are you or someone you know living with chronic pain? Are you looking for the right chronic pain group to get involved with?




